Chinese city hands out £50 COVID-19 vaccine as country steps up battle against virus
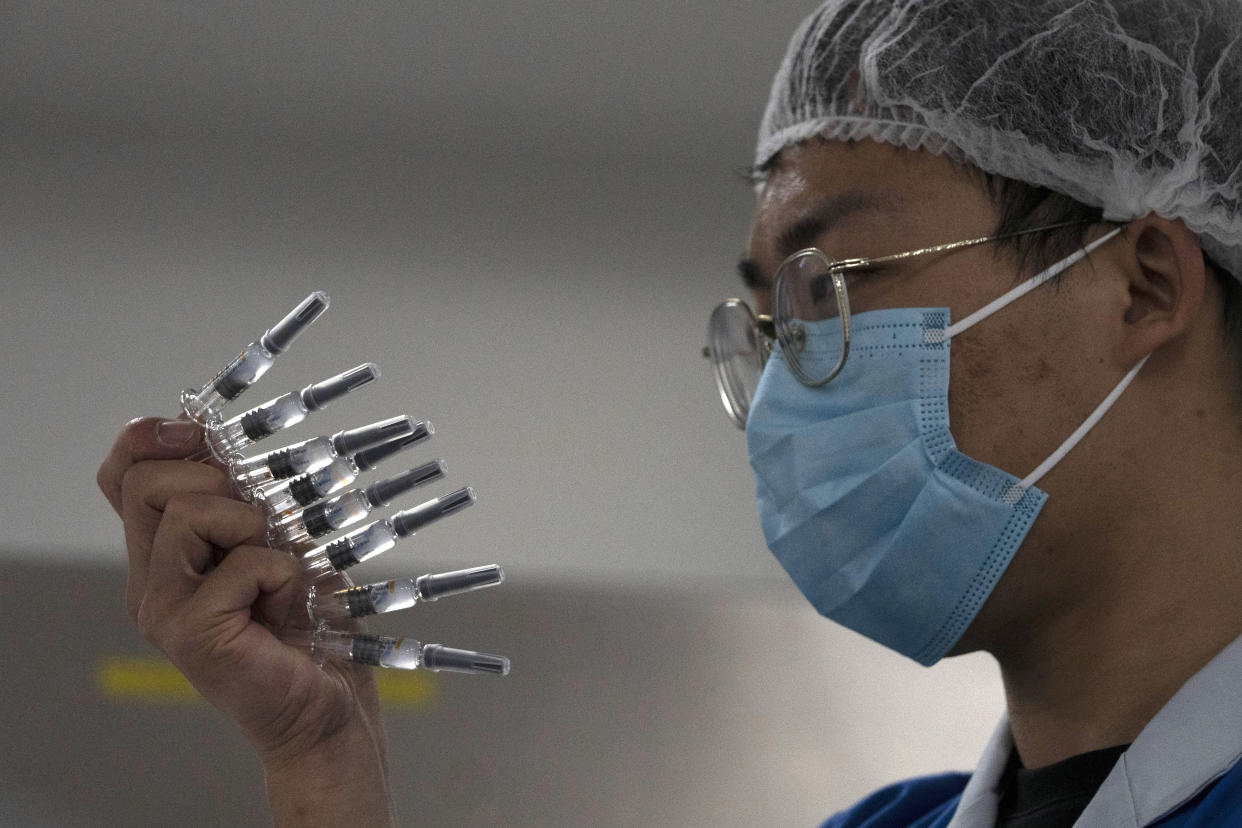
A £50 COVID-19 vaccine is being handed out in a city in China as the country steps up its battle against the disease.
Shaoxing city in Zhejiang province will offer experimental coronavirus vaccines to its residents as China broadens an emergency use programme to people in non-priority groups, the city's health commission said on its WeChat account.
However, it did not name the vaccine, say when inoculation would start, or say how many doses would be offered.
Residents aged between 18 and 59 who are not in priority groups can apply online for inoculation.
Applicants will need to give reasons for wanting the vaccine on their applications and will be charged 400 yuan (£50) for two doses, with an additional inoculation fee of 28 yuan (around £3) per dose, the city said.

The most recent daily figure of new reported cases of COVID-19 in China was 11, out of a combined total of 91,030 since the pandemic began. Reported deaths from the disease stand at 4,739 in the country, according to Johns Hopkins,
Hundreds of thousands of people have already taken experimental COVID-19 vaccines in China since it launched its emergency use programme in July.
The programme was aimed at essential workers and other limited groups of people at higher risk of infection.
Watch: Slim possibility of COVID-19 vaccine by Christmas
Last week, Zhejiang became the first Chinese province to offer the voluntary inoculation to non-priority residents via the emergency use programme, without specifying how many people will be vaccinated.
China approved two COVID-19 inactivated vaccines for a phase I and II combined clinical trial in April, shortly after the disease spread across the globe.
One of those vaccines, CoronaVac, developed by Chinese company Sinovac Biotech, is set to be used in Brazil as part of a national immunisation programme.
Sao Paulo governor Joao Doria described CoronaVac as “the safest [vaccine], the one with the best and most promising rates”.
More than 150 potential vaccines are being developed and tested globally to stop the COVID-19 pandemic, with 42 in human trials, according to the World Health Organization (WHO).
In August, Russia claimed to have the world’s first coronavirus vaccine, with president Vladimir Putin saying it had already been given to his daughter.
However, experts questioned how safe that vaccine was, doubting Putin’s claims that it had “passed all the necessary tests”.
In the UK, several coronavirus jabs are being developed, including by Oxford University and Imperial College London.

Pharmaceutical companies Sanofi and GlaxoSmithKline have also teamed up with the hope of making a COVID-19 vaccine available by the middle of next year.
Meanwhile, volunteers in the UK could be exposed to the coronavirus in controlled settings in a bid to speed up vaccine development.
So-called human challenge trials are expected to start in January 2021, pending approval from regulatory bodies and ethics committees.
Epidemiologist Professor John Edmunds, part of the government’s scientific advisory group Sage, told MPs today it was an “almost certainty” that a vaccine will help to manage the epidemic in the “not-too-distant future” – and possibly by this winter.

He added: “I think these things are moving at pace and of course it’s not just one vaccine being developed but many, many vaccines are being developed across the world.
“The likelihood is that some of these will become available in the not-too-distant future.
“The UK has played a clever game and invested in very many of these and different technologies – I think that’s the right thing to do.
“So I think we here in the UK, we will be in a reasonable position in months.”
Watch: What is long COVID?
Coronavirus: what happened today
Click here to sign up to the latest news and information with our daily Catch-up newsletter

 Yahoo Movies
Yahoo Movies 


