Logistical challenges with the Covid-19 vaccine: Key questions answered
The approval of a Covid-19 vaccine has brought much optimism, but logistical issues may cause problems with getting the jab to care home residents despite them being at the top of the priority list.
What are the possible problems with the Pfizer/BioNTech vaccine and can they be overcome?
The PA news agency has taken a look at some of the latest questions surrounding the first approved coronavirus vaccine.
– When will it arrive and when will the first vaccinations take place?
The first doses of the vaccine – approved by the UK medicines regulator on Wednesday – are due to arrive in the UK on Thursday.
The first #COVID19 vaccine for the UK, developed by @pfizer/@BioNTech_Group has today been given approval for use following a thorough review carried out by the MHRA.
For more info: https://t.co/V481nNZxuL pic.twitter.com/CAD9TVyCtJ
— MHRAgovuk (@MHRAgovuk) December 2, 2020
The UK became the first country in the world to give the go-ahead, with vaccinations expected to start next week.
– Who is at the front of the queue?
Care homes and the elderly are at the top of the list, but logistical issues mean there are difficulties in getting the jab to residents.
The Medicines and Healthcare products Regulatory Agency (MHRA) has not yet given approval to split the vaccine boxes containing 975 doses, meaning some would be wasted if sent to individual residential homes.
Sir Simon Stevens, chief executive of the NHS, has said care home residents will not be able to receive the jab until it is confirmed that batches of the vaccine can be safely divided.
– Does the jab have special storage requirements?
The vaccine needs storage temperatures of minus 70C to minus 80C.
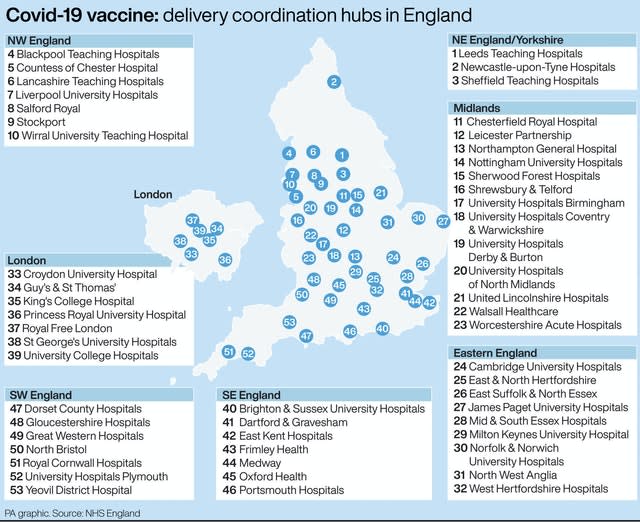
Coronavirus: The 53 vaccine delivery hubs in the UK
Professor Jonathan Van-Tam, the deputy chief medical officer, has said storage requirements for the Pfizer vaccine mean it is a “challenge” to deploy it in care homes.
“If we can get it to care homes then we absolutely will,” he told BBC Breakfast.
He said that if the Oxford University-AstraZeneca vaccine received approval it could be easier to deploy.
– What has Downing Street said?
Number 10 said the NHS is working closely with the regulator to find a way to administer the Pfizer vaccine in care homes.
The Prime Minister’s official spokesman confirmed the first doses would be administered through 50 hospital hubs and would be given to NHS and care home staff and the over-80s.
“The over-80s include those due to come into hospital for routine treatment, so it is obviously possible that that might include some care home residents,” the spokesman said.
“But it is logistically complicated, which is why the NHS are working closely with the MHRA.”
– What have Pfizer and BioNTech said?
They say the vaccine can be sent to care homes as long as it travels for no more than six hours after it leaves cold storage and is then put in a normal fridge at 2C to 8C.
Ben Osborn of Pfizer UK said: “The point I really want to emphasise is, at the point of administration and deployment by the NHS, our vaccine can be stored under normal refrigerated temperatures at 2C-8C for five days.
“And that gives us the flexibility to reach the target populations identified this morning by the JCVI (Joint Committee on Vaccination and Immunisation) over the months ahead.”
Sean Marett, who is chief commercial officer at BioNTech and responsible for distribution, said: “We have stability studies now really supporting the evidence for being able to transport up to six hours at 2C-8C, so you can really take vials from the vaccination centre – one of the large ones – put them in a bag at 2C-8C and take them to the care homes where they can be administered directly to the patients.”
– What about getting the vaccine to elderly people in their own homes?
That seems problematic too. Prof Van-Tam said it would not be viable to get individual doses to elderly people unable to leave their homes, and suggested they may need to wait for the Oxford/AstraZeneca vaccine.
He said he is hoping for approval by Christmas for the Oxford vaccine, which is “much easier to split into smaller quantities and probably can go into people’s homes”.
– How likely is it that elderly people will have to wait until another vaccine is approved?
Sir Simon said the NHS is “raring to go” to vaccinate people in care homes, hopefully this month.
“If the MHRA, the independent regulator, as we expect they will, give approval for a safe way of splitting these packs of 975 doses, then the good news is we will be able to start distributing those to care homes,” he said.
But experts have suggested care home residents and elderly people who cannot leave their own homes may have to wait for other vaccines to be approved.

 Yahoo Movies
Yahoo Movies 
